Isothermal titration calorimetryIsothermal titration calorimetry ( ITC) is a physical technique used to determine the parameters of interactions in solution. It is most often used to study the binding of small molecules (such as medicinal compounds) to larger (, etc.).It consists of two cells which are enclosed in an jacket. The compounds to be studied are placed in the sample cell, while the other cell, the reference cell, is used as a control and contains the buffer in which the sample is dissolved.
Energetically favorable binding reactions have negative free energy values, ΔG = RTlnK D. ΔG, has two energetic components, enthalpy (ΔH) and entropy (ΔS) and their contributions are expressed as:ΔG = ΔH -TΔS.In an ITC experiment, ΔH of binding is measured directly. The ITC200 microcalorimeter has two cell: one contains water and acts as a reference cell, the other contains the sample. The microcalorimeter needs to keep these two cells at exactly the same temperature during the course of an experiment. Heat sensing devices detect temperature difference between the cells when binding occurs in the sample cell and give feedback to the heaters, which compensate for this difference and return the cells to equal temperature. Data collectionTo perform an experiment, the calorimeter is set to the experimental temperature. The sample cell is filled and the injection syringe is loaded with ligand.
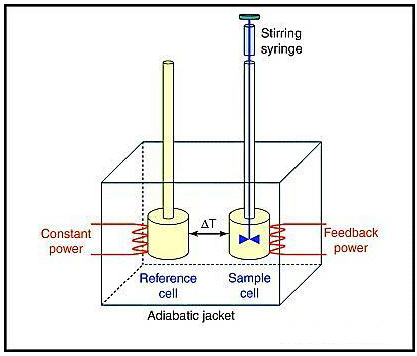
Isothermal Titration Calorimetry (ITC) is a standard technique designed to characterize intermolecular interactions. AFFINImeter has been designed to really squeeze the information that can be obtained from ITC experiments, ranging from kinetic interaction constants to reaction mechanisms and thermodynamic parameters of relatively complex reactions. In this context, Isothermal Titration Calorimetry (ITC) is a powerful technique for the characterization of macromolecular interactions. ITC determines the heat change that occurs upon the binding of two molecules. Heat can be absorbed (endothermic reaction) or released (exothermic reaction).
The syringe is inserted into the sample cell and and a series of small aliquots of ligand are injected into the sample solution, while stirring. If there is a binding of the ligand to the sample, heat changes of a few millionths of a degree Celsius are detected and measured. The microcalorimeter measures all heat released until the binding reaction has reached equilibrium. The instrument records the differential power (in µcal/sec) applied to the sample cell vs. The reference cell to keep the two cells at the same temperature. ResultsThe example below shows an exothermic reaction, which means the sample cell becomes warmer than the reference cell and causes a downward peak in the signal.
As the temperature of the two cells equilibrate, the signal returns to its starting position. As the molar ratio between the ligand and sample increases, the sample becomes saturated and fewer injected ligand molecules bind and the heat change decreases. By integrating the area of the injection peaks and plotting molar ratio vs. ∆H (kcal/mol), an ITC binding curve can be fit for binding affinity (K A = 1/K D), reaction stoichiometry (n), enthalpy (∆H) and entropy (ΔS). The relative contribution of enthalpy and entropy to the binding energetics can provide insights into molecular mechanism: ∆H is an indication of changes in hydrogen bonding and van der Waals interactions, and ∆S is an indication of changes in hydrophobic effects and conformational changes.Image from Malvern Instruments.
